소개

Prof. 고제상
Tel: 02-3290-3445(office)
Tel: 02-3290-3923(Lab.)
E-mail: jesangko@korea.ac.kr
- About Professor
- Curriculum Vitae
- Publication
- Research
- Teaching
- Lab Members
Profile
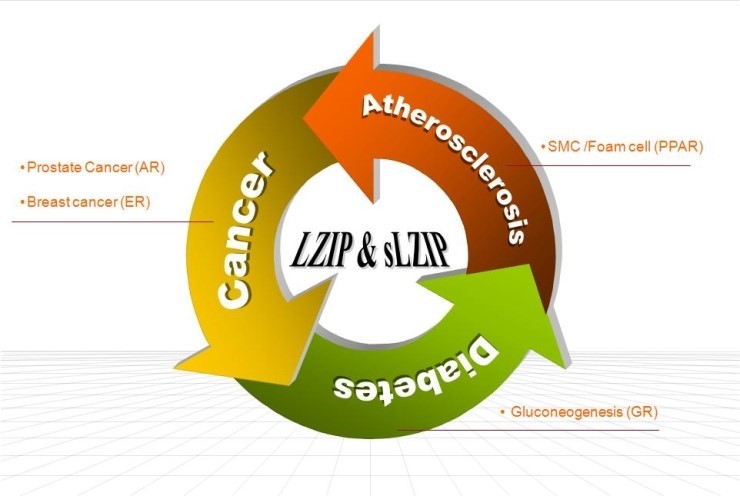
생체 세포의 여러 가지 생명 현상, 즉 세포의 증식과 분화, 대사, 분비, 유전자 발현 등은 세포들 간의 유기적인 정보 교환과 외부 신호에 의한 세포 내에서의 일련의 신호전달 과정을 통해 조절되고 유지된다. 특히, 생체미세환경 내에 존재하는 세포들의 만성적 염증반응에 관련된 신호전달 체계의 불균형은 암과 동맥경화와 같은 비정상적 만성 염증성 질환을 유발한다.
최근에 본 연구실에서 발견한 small leucine zipper protein (sLZIP)과 그 isoform 인 LZIP은 전사인자로써 혈관평활근세포의 이동과 증식을 조절하고, 거품세포 형성에 관여하며, 혈관벽 및 동맥경화반에서 강하게 발현되어 있어 동맥경화의 발병과정에서 중요한 역할을 할 것으로 예상된다. 특히, 전사인자 PPARγ의 전사활성과 MMP 효소들의 발현 및 활성을 조절하여 혈관내 염증성 유도물질들을 분비함으로써 동맥경화를 유도하는 것으로 생각되고 있다. 본 연구실은 sLZIP과 LZIP을 중심으로 동맥경화에 관련된 단백질들의 기능을 분자적 수준에서 이해하고, 이 단백질들을 질병 치료를 위한 target molecule로 제시하여 동맥경화 발병기전 규명과 치료제 개발에 공헌하고자 한다.
안드로겐 수용체 (AR), 에스트로겐 수용체 (ER), 글루코콜티코이드 수용체 (GR) 등의 핵 수용체는 개체의 발생 및 분화, 생식, 염증반응, 항상성 유지 등 개체유지에 중요한 다양한 과정에서 기능을 하며, 이러한 핵 수용체가 관련된 신호전달체계의 이상은 전립선암, 유방암, 대사성 질환 등 많은 질병과 연관되어 있다.
최근 본 연구실은 AR과 ER에 결합하고 전사활성을 조절하여 전립선암과 유방암의 발병과정에서 중요한 기능을 하는 단백질을 발견하였다. 본 연구실은 전립선암 및 유방암 세포에서 이 단백질에 의한 핵 수용체 표적 단백질들의 발현 조절 작용을 규명하고, 형질전환 동물모델을 이용하여 이를 검증함으로써 전립선암 및 유방암 진단/치료를 위한 바이오마커를 개발하려고 한다. 또한, GR과 결합하여 GR의 전사활성을 조절하는 단백질에 의한 당 대사 조절 기전을 규명하여 대사성 질환 관련성 및 분자세포생물학적 치료방법 개발에 대한 표적단백질로서 가능성을 제시하고자 한다.
Curriculum Vitae
Education
Ph.D.
(1993) Biological Sciences, University of Rhode Island, RI, USA
MS
(1987) Food Biosciences and Technology, Korea University, Seoul, Korea
BS
(1985) Food Biosciences and Technology, Korea University, Seoul, Korea
Professional Experiences
- 2009. 3 - present : Professor, Korea University, Seoul, Korea
- 2005. 3 - 2009. 2 : Associate Professor, Korea University, Seoul, Korea
- 1999. 12 - 2005. 2 : Assistant Professor, University of Ulsan College of Medicine, Seoul, Korea
- 1997. 5 - 1999. 11 : Research Fellow, Dana-Farber Cancer Institute/Harvard Medical School, MA, USA
- 1995. 1 - 1997. 4 : Research Fellow, Harvard Medical School, MA, USA
- 1994. 1 - 1994. 12 : Post-doc, University of Rhode Island, RI, USA
Publication
Suhyun Kim, Minseok Oh, Minsoo Kang, and Jesang Ko. (2022) Small leucine zipper protein functions as a modulator for metabolic reprogramming of colorectal cancer cells by inducing nutrient stress-mediated autophagy. Cellular and Molecular Life Sciences. 79(9).
Suhyun Kim, Minsoo Kang, and Jesang Ko. (2021) Small leucine zipper protein promotes the metastasis of castration-resistant prostate cancer through transcriptional regulation of matrix metalloproteinase-13. Carcinogenesis. 42(8), 1089-1099.
Minsoo Kang, Sun Kyoung Han, Suhyun Kim, Sungyeon Park, Yerin Jo, Hyeryung Kang and Jesang Ko. (2021) Role of small leucine zipper protein in hepatic gluconeogenesis and metabolic disorder. J Mol Cell Biol. 13(5), 361-373.
-
-
Sungyeon Park, Minsoo Kang, Suhyun Kim, Hyoung-Tae An, Jan Gettemans, and Jesang Ko. (2020) α-Actinin-4 promotes the progression of prostate cancer through the Akt/GSK-3β/β-catenin signaling pathway. Frontiers in Cell and Developmental Biology. 8, 588544.
-
-
Seukun Kim*, Sungyeon Park*, Minsoo Kang, and Jesang Ko. (2020) The role of small leucine zipper protein in osteoclastogenesis and its involvement in bone remodeling. BBA-Mol Cell Res. 1867(11), 118827. *co-first authors.
-
Jaeyeon Jung*, Suhyun Kim*, Hyoung-Tae An, and Jesang Ko (2020) α-Actinin-4 regulates cancer stem cell properties and chemoresistance in cervical cancer. Carcinogenesis. 41(7), 940-949. *co-first authors.
-
Minsoo Kang, Suhyun Kim, and Jesang Ko. (2019) Roles of CD133 of microvesicle formation and oncoprotein trafficking in colon cancer. FASEB J. 33(3), 4248-4260.
-
Yunhee Hwang*, Hyoung-Tae An*, Minsoo Kang, and Jesang Ko. (2018) Role of 14-3-3β and γ in regulation of the glucocorticoid receptor transcriptional activation and hepactic gluconeogenesis. Biochem Biophys Res Commun. 501(3), 800-806. *co-first authors.
-
A-ram Kang, Hyoung-Tae An, Jesang Ko, Eui Ju Choi, and Seongman Kang. (2017) Ataxin-1 is involved in tumorigenesis of cervical cancer cells via the EGFR-RAS-MAPK signaling pathway. Oncotarget. 8(55), 94606-94618.
-
Dong-Min Yu, Seung Hee Jung, Hyoung-Tae An, Sungsoo Lee, Jin Hong, Jun Sub Park, Hyun Lee, Hwayeon Lee, Myeong-Suk Bahn, Hyung Chul Lee, Na-Kyung Han, Jesang Ko, Jae-Seon Lee, Young-Gyu Ko. (2017) Caveolin-1 deficiency induces premature senescence with mitochondrial dysfunction. Aging Cell, 16(4), 773-784.
-
Juyeon Jeong*, Sodam Park*, Hyoung-Tae An, Minsoo Kang, and Jesang Ko. (2017) Small leucine zipper protein functions as a negative regulator of estrogen receptor α in breast cancer. PLoS One 12(6), e0180197. *co-first authors.
-
-
Minsoo Kang, Jeonghan Kim, Hyoung-Tae An, and Jesang Ko. (2017) Human leucine zipper protein promotes hepatic steatosis via induction of apolipoprotein A-IV. FASEB J. 31(6) 2548-2561.
-
A-ram Kang, Hyoung-Tae An, Jesang Ko, and Seongman Kang. (2017) Ataxin-1 regulates epithelial-mesenchymal transition of cervical cancer cells. Oncotarget. 8(11), 18248-18259.
-
Beom-Chan Kim, Hyun-Jung Hwang, Hyoung-Tae An, Hyun Lee, Jun-Sub Park, Jin Hong, Jesang Ko, Chungho Kim, Jae-Seon Lee, Young-Gyu Ko. (2016) Antibody neutralization of cell-surface gC1qR/HABP1/SF2-p32 prevents lamellipodia formation and tumorigenesis . Oncotarget. 7(31), 49972-49985.
-
-
Hyoung-Tae An, Seungmin Yoo and Jesang Ko. (2016) α-Actinin-4 induces the epithelial-to-mesenchymal transition and tumorigenesis via regulation of Snail expression and β-catenin stabilization in cervical cancer. Oncogene. 35(45), 5893-5904.
-
Ok Hyun Park, Joori Park, Mira Yu, Hyoung-Tae An, Jesang Ko, and Yoon Ki Kim. (2016) Identification and molecular characterization of cellular factors required for glucocorticoid receptor-mediated mRNA decay. Genes and Development. 30(18), 2093-2105.
- Sodam Park*, Seungmin Yoo*, Jeonghan Kim, Hyoung-Tae An, Minsoo Kang, and Jesang Ko. (2015) 14-3-3β and γ differentially regulate peroxisome proliferator activated receptor γ2 transactivation and hepatic lipid metabolism . BBA-Gene Regul Mech., 1849(10), 1237-1247. *co-first authors.
-
Hana Cho, Ok Hyun Park, Joori Park, Incheol Ryu, Jeonghan Kim, Jesang Ko, and Yoon Ki Kim. (2015) Glucocorticoid receptor interacts with PNRC2 in a ligand-dependent manner to recruit UPF1 for rapid mRNA degradation. Proc Natl Acad Sci, USA. 112(13), E1540-1549.
-
Yoonseo Kim*, Jeonghan Kim*, Sung-Wuk Jang, and Jesang Ko. (2015) The role of sLZIP in negative regulation of cyclin D3-mediated androgen receptor transactivation and its involvement in prostate cancer. Oncogene. 34(2), 226-236. *co-firtst authors.
-
Jeonghan Kim, and Jesang Ko. (2014) Human sLZIP promotes atherosclerosis via MMP-9 transcription and vascular smooth muscle cell migration . FASEB J. 28(11), 5010-5021.
-
Jeonghan Kim, and Jesang Ko. (2014) A novel PPARγ2 modulator sLZIP controls the balance between adipogenesis and osteogenesis during mesenchymal stem cell differentiation . Cell Death & Differentiation. 21(10), 1642-1655.
-
Jeonghan Kim, Sung-Wuk Jang, Eunsoo Park, Minseok Oh, Sodam Park, and Jesang Ko. (2014) The role of heat shock protein 90 in migration and proliferation of vascular smooth muscle cells in the development of atherosclerosis . J Mol. Cell Cardiol. 72(C), 157-167.
-
Eunsoo Park, Hyereen Kang, Jeonghan Kim, and Jesang Ko. (2014) The role of sLZIP in transcriptional regulation of c-Jun and involvement in migration and invasion of cervical cancer cells . Cell Physiol. Biochem. 33(1), 151-164.
-
Hyoung-Tae An, Jeonghan Kim, Seungmin Yoo, and Jesang Ko. (2014) Small leucine zipper protein (sLZIP) negatively regulates skeletal muscle differentiation via interaction with α-actinin-4 . J Biol Chem. 289(8), 4969-4979.
- Yoon Suk Kim, Jeonghan Kim, Chi A Yi, Jesang Ko, Yong Serk Park, Seung-Jae Lee.(2012) Macromolecular Crowding Enhances Interaction of α-synuclein with Vesicles. J Exp Biomed Sci. 18(4), 329-337.
-
Hyereen Kang, Jesang Ko, and Sung-Wuk Jang. (2012) The role of annexin A1 in expression of matrix metalloproteinase-9 and invasion of breast cancer cells. Biochem Biophys Res Commun. 423(1), 188-194.
-
-
Hyereen Kang, Minjae Lee, Kyung-chul Choi, Dong-Myoung Shin, Jesang Ko and Sung-Wuk Jang. (2012) N-(4-hydroxyphenyl)retinamide inhibits breast cancer cell invasion through suppressing NF-kB activation and inhibiting matrix metalloproteinase-9 expression . J Cell Biochem. 113(9), 2845-2855.
-
-
Eun Ju Yang, Eugene Choi, Jesang Ko, Dong-Hee Kim, Ji-Sook Lee, and In Sik Kim. (2012) Differential effect of CCL2 on constitutive neutrophil apoptosis between normal and asthmatic subjects. J Cell Physiol. 227(6), 2567-2577.
-
-
Ho Joong Sung, Jeonghan Kim, Yoonseo Kim, Sung-Wuk Jang, and Jesang Ko. (2012) N-acetyl cysteine suppresses the foam cell formation that is induced by oxidized low density lipoprotein via regulation of gene expression. Mol Biol Rep. 39(3), 3001-3007.
-
Ho Joong Sung, Yoonseo Kim, Hyereen Kang, Jae Woong Sull, Yoon Suk Kim, Sung-Wuk Jang, and Jesang Ko. (2012) Inhibitory effect of Trolox on the migration and invasion of human lung and cervical cancer cells. Int J Mol Med. 29(2), 245-251.
-
-
Soon Young Jang, Sung-Wuk Jang, and Jesang Ko. (2012) Regulation of ADP-ribosylation factor 4 expression by small leucine zipper protein and involvement in breast cancer cell migration. Cancer Lett. 314(2), 185-197.
-
Hyereen Kang, Sung-Wuk Jang, and Jesang Ko. (2011) Human leucine zipper protein sLZIP induces migration and invasion of cervical cancer cells via expression of matrix metalloproteinase-9. J Biol Chem. 286(49), 42072-42081.
-
-
Yoonseo Kim, Hyungjin Kim, Sung-Wuk Jang, and Jesang Ko. (2011) The role of 14-3-3β in transcriptional activation of estrogen receptor α and its involvement in proliferation of breast cancer cells. Biochem Biophys Res Commun. 414(1), 199-204.
-
-
Yoonseo Kim, Hyereen Kang, Sung-Wuk Jang, and Jesang Ko. (2011) Celastrol inhibits breast cancer cell invasion via suppression of NF-ĸB-mediated matrix metalloproteinase-9 expression. Cell Physiol Biochem. 28(2), 175-184.
-
-
Soon Young Jang, Sung-Wuk Jang, and Jesang Ko. (2011) Celastrol inhibits the growth of estrogen positive human breast cancer cells through modulation of estrogen receptor α. Cancer Lett. 300(1), 57-65.
- Jung-A Choi, Jin-Wook Lee, Hyunju Kim, Eun-Young Kim, Ji-Min Seo, Jesang Ko, and Jae-Hong Kim. (2010) Pro-survival of estrogen receptor -negative breast cancer cells is regulated by a BLT2-reactive oxygen species-linked signaling pathway. Carcinogenesis. 31(4), 543-551.
- Jeonghan Kim, Yoon Suk Kim, and Jesang Ko. (2010) CKβ8/CCL23 and its isoform CKβ8-1 induce up-regulation of cyclins via the G(i)/G(o) protein/PLC/PKCδ/ERK leading to cell-cycle progression. Cytokine. 50(1), 42-49.
- Jeonghan Kim, Yoon Suk Kim, and Jesang Ko. (2010) CKβ8/CCL23 induces cell migration via the Gi/Go protein/PLC/PKCδ/NF-κB and is involved in inflammatory responses. Life Sci. 86, 300-308.
- Yoon Suk Kim, Hyoung Tae An, Jeonghan Kim, and Jesang Ko. (2009) Effects of protein kinase Cδ and phospholipase C-γ1 on monocyte chemoattractant protein-1 expression in taxol-induced breast cancer cell death. Int. J. Mol. Med. 24 (6), 853-858.
- Hyereen Kang, Yoon Suk Kim, and Jesang Ko. (2009) A novel isoform of human LZIP negatively regulates the transactivation of the glucocorticoid receptor. Mol Endo. 23 (11):1746-1757.
- Geun Hyang Kim, Keunhee Park, Seon-Yong Yeom, Kyung Jin Lee, Gukhan Kim, Jesang Ko , Dong-Kwon Rhee, Young Hoon Kim, Hye Kyung Lee, Hae Won Kim, Goo Taeg Oh, Ki-Up Lee, Jae W. Lee, and Seung-Whan Kim (2009) Characterization of ASC-2 as an antiatherogenic transcriptional coactivator of liver X receptors in macrophages. Mol Endo. 23 (7), 966-974.
- Byong Wook Min, Chang Gun Kim, Jesang Ko, Yoongho Lim, Young Han Lee, and Soon Young Shin. (2008) Transcription of the protein kinase C-δ gene is activated by JNK through c-Jun and ATF2 in response to the anticancer agent doxorubicin . Exp. Mol. Med. 40 (6), 699-708.
- Yoon Suk Kim, Jeonghan Kim, Yoonseo Kim, Young Han Lee, Jae-Hong Kim, Seung-Jae Lee, Soon Young Shin, and Jesang Ko. (2008) The role of calpains in ligand-induced degradation of the glucocorticoid receptor. Biochem. Bioph. Res. Co. 374 (2), 373-377.
- Ho Joong Sung, Yoon Suk Kim, Hyereen Kang, and Jesang Ko. (2008) Human LZIP induces monocyte CC chemokine receptor 2 expression leading to enhancement of monocyte chemoattractant protein 1/CCL2-induced cell migration. Exp. Mol. Med. 40 (3), 332-338.
- Sang Min Jung, KiBeom Lee, Joung Wook Lee, Hong Namkoong, Hyun Kee Kim, Sanghee Kim, Hae Ri Na, Seon-Ah Ha, Jae-Ryong Kim, Jesang Ko, and Jin Woo Kim. (2008) Both plasma retinol-binding protein and haptoglobin precursor allele 1 in CSF: candidate biomarkers for the progression of normal to mild cognitive impairment to Alzheimer's disease. Neurosci. Lett. 436 (2), 153-157.
- Sung-Wuk Jang, Yoon Suk Kim, Yoon Rim Kim, Ho Joong Sung, and Jesang Ko. (2007) Regulation of human LZIP expression by NF-kappaB and its involvement in monocyte cell migration induced by Lkn-1. J. Biol. Chem. 282(15), 11092-11100.
- Sung-Wuk Jang, Yoon Suk Kim, Young Han Lee, and Jesang Ko. (2007) Role of human LZIP in differential activation of the NF-kappaB pathway that is induced by CCR1-dependent chemokines. J. Cell. Physiol. 211(3), 630-637.
- Jesang Ko, Chi-Young Yun, Ji-Sook Lee, Joo-Hwan Kim, and In Sik Kim. (2007) p38 MAPK and ERK activation by 9-cis-retinoic acid induces chemokine receptors CCR1 and CCR2 expression in human monocytic THP-1 cells. Exp. Mol. Med. 39(2), 129-138.
- Soon Young Shin, Byeong Hyeok Choi, Jesang Ko, Se Hyun Kim, Yong Sik Kim, and Young Han Lee. (2006) Clozapine, a neuroleptic agent, inhibits Akt by counteracting Ca2+/calmodulin in PTEN-negative U-87MG human glioblastoma cells. Cell. Signal. 18(11), 1876-1886.
- Jesang Ko, Chi-Young Yun, Ji-Sook Lee, Dong-Hee Kim, Ji Eun Yuk, and In Sik Kim. (2006) Differential regulation of CC chemokine receptors by 9-cis retinoic acid in the human mast cell line, HMC-1. Life Sci. 79(13), 1293-1300.
- Seung Min Shin, Yeun Jun Chung, Seong Tack Oh, Hae Myung Jeon, Lae Jeong Hwang, Hong Namkoong, Hyun Kee Kim, Goang Won Cho, Soo Young Hur, Tae Eung Kim, Youn Soo Lee, Yong Gyu Park, Jesang Ko, and Jin Woo Kim. (2006) HCCR-1-interacting molecule "deleted in polyposis 1" plays a tumor-suppressor role in colon carcinogenesis. Gastroenterology, 130 (7), 2074-2086.
Sang Seol Jung, Hyung Soon Park, Insong James Lee, Hong Namkoong, Seung Min Shin, Goang Won Cho, Seon-Ah Ha, Yong Gyu Park, Youn Soo Lee, Jesang Ko, and Jin Woo Kim. (2005) The HCCR oncoprotein as a biomarker for human breast cancer. Clin. Cancer Res. 11, 7700-7708.
In Sik Kim, Sung-Wuk Jang, Ho Joong Sung, Ji-Sook Lee, and Jesang Ko. (2005) Differential CCR1-mediated chemotaxis signaling induced by human CC chemokine HCC-4/CCL16 in HOS cells. FEBS Lett. 579, 6044-6048.
Yoon Suk Kim, Sung-Wuk Jang, Ho Joong Sung, Hye Jin Lee, In Sik Kim, Doe Sun Na, and Jesang Ko. (2005) Role of 14-3-3η as a positive regulator of the glucocorticoid receptor transcriptional activation. Endocrinology. 146, 3133-3140.
Jungsu Hwang, Kyung-No Son, Chan Woo Kim, Jesang Ko, Doe Sun Na, Byoung S. Kwon, Yong Song Gho, and Jiyoung Kim. (2005) Human CC chemokine CCL23, a ligand for CCR1, induces endothelial cell migration and promotes angiogenesis. Cytokine. 30, 254-263.
Ki Hoon Han, Jewon Ryu, Kyung Hee Hong, Jesang Ko, Youngmi Kim Pak, Jae-Bum Kim, Seong Wook Park, Jae Joong Kim. (2005) HMG-CoA Reductase Inhibition Reduces Monocyte CC Chemokine Receptor 2 Expression and Monocyte Chemoattractant Protein-1–Mediated Monocyte Recruitment In Vivo. Circulation. 111, 1439-1447.
Sang Min Lee, Hwayean Shin, Sung-Wuk Jang, Jung-Jae Shim, In-sung Song, Kyung-No Son, Jungsu Hwang, Yong-Hyun Shin, Hong-Hee Kim, Chong-Kil Lee, Jesang Ko, Doe Sun Na, Byoung S. Kwon, and Jiyoung Kim. (2004) PLP2/A4 interacts with CCR1 and stimulates migration of CCR1-expressing HOS cells. Biochem. Bioph. Res. Co. 324, 768-772.
Ho Joong Sung, Yoon Suk Kim, In Sik Kim, Sung-Wuk Jang, Yoon Rim Kim, Doe Sun Na, Ki Hoon Han, Byung Gil Hwang, Dong Suk Park and Jesang Ko. (2004) Proteomic analysis of differential protein expression in neuropathic pain and electroacupuncture treatment models. Proteomics. 4, 2805-2813.
Jungsu Hwang, Chan Woo Kim, Kyung-No Son, Kyu Yeon Han, Kyung Hee Lee, Hynda K. Kleinman, Jesang Ko, Doe Sun Na, Byoung S. Kwon, Yong Song Gho, and Jiyoung Kim. (2004) Angiogenic activity of human CC chemokine CCL15 in vitro and in vivo. FEBS Lett. 570, 47-51.
Soon Young Shin, Chang Gun Kim, Jesang Ko, Do Sik Min, Jong-Soo Chang, Yoon Suk Kim, Motoi Ohba, Toshio Kuroki, Choi young Bong, Young-Ho Kim, Doe Sun Na, Jin Woo Kim, and Young Han Lee. (2004) Transcriptional and post-transcriptional regulation of the PKCδ gene by etoposide in L1210 murine leukemia cells: Implication of PKCδ autoregulation. J. Mol. Biol. 340, 681-693.
In Sik Kim, Yoon Suk Kim, Sung-Wuk Jang, Ho Joong Sung, Ki Hoon Han, Doe Sun Na, and Jesang Ko. (2004) Differential effects of 9-cis retinoic acid on expression of CC chemokine receptors in human monocytes. Biochem. Pharm. 68, 611-620.
Ki Hoon Han, Kyung Hee Hong, Jesang Ko, Kyong Suk Rhee, Myeong Ki Hong, Jae Joong Kim, You Ho Kim, Seung Jung Park. (2004) Lysophosphatidylcholine up-regulates CXCR4 chemokine receptor expression in human CD4 T cells. J. Leukoc. Biol. 76, 195-202.
Ki Hoon Han, Kyung-Hee Hong, Jae-Hyeong Park, Jesang Ko, Duk-Hyun Kang, Kee-Joon Choi, Myeong-Ki Hong, Seong-Wook Park, Seung-Jung Park. (2004) C-reactive protein promotes monocyte chemoattractant protein-1-mediated chemotaxis through upregulating CC chemokine receptor 2 expression in human monocytes. Circulation. 109, 2566-2571.
Je-Won Ryu, Kyung Hee Hong, Jin Hee Maeng, Jae-Bum Kim, Jesang Ko, Joong Yeol Park, Ki-Up Lee, Myeong Ki Hong, Seong Wook Park, You Ho Kim, Ki Hoon Han. (2004) Overexpression of uncoupling protein 2 in THP1 monocytes inhibits β2 integrin-mediated firm adhesion and transendothelial migration. Arterioscler. Thromb. Vasc. Biol. 24, 864-870.
Jesang Ko, Sung-Wuk Jang, Yoon Suk Kim, In Sik Kim, Ho Joong Sung, Hong-Hee Kim, Joong-Yeol Park, Young Han Lee, Jiyoung Kim, and Doe Sun Na. (2004) Human LZIP binds to CCR1 and differentially affects the chemotactic activities of CCR1-dependent chemokines. FASEB J.
Jesang Ko, Seung Min Shin, Young Mi Oh, Youn Soo Lee, Zae Young Ryoo, Young Han Lee, Doe Sun Na, and Jin Woo Kim. (2004) Transgenic mouse model for breast cancer: Induction of breast cancer in novel oncogene HCCR-2 transgenic mice. Oncogene. 23, 1950-1953.
Moon Suk Kim, Sung-Su Kim, Sang Taek Jung, Jung-Young Park, Han-Wook Yoo, Jesang Ko, Katalin Csiszar, Sang-Yun Choi, and Youngho Kim. (2003) Expression and purification of enzymatically active forms of the human lysyl oxidase-like protein 4. J. Biol. Chem.
Yoon Suk Kim, Jesang Ko, In Sik Kim, Sung-Wuk Jang, Ho Joong Sung, Hye Jin Lee, Si Yeol Lee, Youngho Kim, and Doe Sun Na. (2003) PKCδ-dependent cleavage and nuclear translocation of annexin A1 by phorbol 12-myristate 13-acetate. Eur. J. Biochem. 270, 4089-4094.
Jesang Ko, Young Han Lee, Seung Yong Hwang, Youn Soo Lee, Seung Min Shin, Jae Hoon Hwang, Jin Kim, Yong Wook Kim, Sung-Wuk Jang, Zae Young Ryoo, In-Kyung Kim, Sung Eun Namkoong, and Jin Woo Kim. (2003) Identification and differential expression of novel human cervical cancer oncogene HCCR-2 in human cancers and its involvement in p53 stabilization. Oncogene. 22, 4679-4689.
In Sik Kim, Yong Suk Ryang, Yoon Suk Kim, Sung-Wuk Jang, Ho Joong Sung, Young Han Lee, Jiyoung Kim, Doe Sun Na, and Jesang Ko. (2003) Leukotactin-1-induced ERK activation is mediated via Gi/Go protein/PLC/PKCδ/Ras cascades in HOS cells. Life Sci. 73, 447-459.
Jesang Ko, Ki Sung Ryu, Young Han Lee, Doe Sun Na, Yoon Suk Kim, Young Mi Oh, In Sik Kim, and Jin Woo Kim. (2002) Human secreted frizzled-related protein is down-regulated and induces apoptosis in human cervical cancer. Exp. Cell Res. 280, 280-287.
Soon Young Shin, Jesang Ko, Jong-Soo Chang, Do Sik Min, Chan Choi, Sun Sik Bae, Myung Jong Kim, Dae Sung Hyun, Jung-Hye Kim, Mi Young Han, Young-Ho Kim, Yong Sik Kim, Doe Sun Na, Pann-Ghill Suh, and Young Han Lee. (2002) Negative regulatory role of overexpression of PLC1 in the expression of early growth response 1 gene in rat 3Y1 fibroblasts. FASEB J. 16, 1504-1514.
Jesang Ko, Doe Sun Na, Young Han Lee, Soon Young Shin, Ji Hoon Kim, Byung Gil Hwang, Byung-Il Min, and Dong Suk Park. (2002) cDNA microarray analysis of differential gene expression in neuropathic pain and electroacupuncture treatment models. Journal of Biochemistry and Molecular Biology. 35(4), 420-427.
Jesang Ko, In Sik Kim, Sung-Wuk Jang, Young Han Lee, Soon Young Shin, Do Sik Min, and Doe Sun Na. (2002) Leukotactin-1-induced chemotaxis signaling through CCR1 in HOS cells. FEBS Lett. 515, 159-164.
Seung-Wook Kim, Hae Jin Rhee, Jesang Ko, Yeo Jeong Kim, Hyung Gu Kim, Jai Myung Yang, Eung Chil Choi, and Doe Sun Na. (2001) Inhibition of cytosolic phospholipase A2 by annexin-I: Specific interaction model and mapping of the interaction site. J. Biol. Chem. 276, 15712-15719.
Soon Young Shin, Seong-Yong Kim, Jung-Hye Kim, Do Sik Min, Jesang Ko, Ung-Gu Kang, Yong Sik Kim, Taeg Kyu Kwon, Young Ho Kim, and Young Han Lee. (2001) Induction of early growth response-1 gene expression by calmodulin antagonist trifluoperazine through the activation of Elk-1 in human fibrosarcoma HT1080 cells. J. Biol. Chem. 276, 7797-7805.
Seung-Wook Kim, Jesang Ko, Jae Hong Kim, Eung Chil Choi, and Doe Sun Na. (2001) Differential effects of annexins I, II, III, and V on cytosolic phospholipase A2 activity: Specific interaction model. FEBS Lett. 489, 243-248.
-
Research
Research Area
Nuclear transcription factors, including androgen receptor (AR), estrogen receptor (ER), and glucocorticoid receptor (GR), play critical roles in the development of prostate and breast cancers and inflammatory responses. We found that a novel protein binds to AR and ER, and negatively regulates the transcriptional activities of AR and ER. We are trying to characterize the role of this protein in prostate and breast cancers and identify biomarkers of these cancers. We are also interested in GR transcriptional regulators that are involved in glucose metabolism. Atherosclerosis is a chronic inflammatory response of the vascular wall. We are attempting to understand the role of a novel transcription factor, the human small leucine zipper protein (sLZIP), in vascular smooth muscle cell (VSMC) migration and atherosclerosis progression. We found that sLZIPOE/+;LDLR-/- mice fed a high cholesterol diet exhibit enhanced arterial plaque formation and increased VSMC migration. We are studying whether sLZIP can be used as a therapeutic target molecule for treatment of atherosclerosis.
Teaching
Cell Biology 1 (LIST201)
Cell Biology 2 (LIST202)
Cancer Biology (LIST401)
Cancer Biology 1 (CEL979)
Cardiovascular Biology (CEL554)
Lab Members
Present
- Sungyeon Park
- Ph.D
- sungyeon10@korea.ac.kr
- Sunyoung Lee
- MS/Ph.D Student
- christy0925@kribb.re.kr
- Sujin Kwon
- MS student
- kwsu9914@korea.ac.kr
Former
- Yoon Suk Kim
- Professor, Yonsei Univ.
- yoonsukkim@yonsei.ac.kr
- In Sik Kim
- Professor, Eulji Univ.
- orientree@eulji.ac.kr
- Sung-Wuk Jang
- Associate Professor, Univ. of Ulsan College of Medicine
- swjang@amc.seoul.kr
- Ho Joong Sung
- Associate Professor, Eulji Univ.
- hjsung@eulji.ac.kr
- Hyereen Kang
- Research Professor, Yonsei Univ. College of Medicine
- frejya1052@gmail.com
- Geun Hyang Kim
- Scientist, Regeneron Pharmaceuticals, USA
- hyang.kim@regeneron.com
- Soon Young Jang
- Post-doc, Korea Univ. College of Medicine
- agct98@gmail.com
- Yoonseo Kim
- Post-doc, NIH, USA
- kys2117@hanmail.net
- Hyung Jin Kim
- Samsung Electronics Co.
- abimael@naver.com
- Sun Kyoung Han
- Seoul Nat. Univ. Hospital
- luvsun@korea.ac.kr
- Yunhee Hwang
- Thermo Fisher Scientific
- hyh07@hanmail.net
- Eunsoo Park
- Researcher
- parkespink@naver.com
- Sodam Park
- Amorepacific
- cookiestar@korea.ac.kr
- Minseok Oh
- Samsung Biologics
- daeguoms@korea.ac.kr
- Jeonghan Kim
- Assistant professor, The Catholic University of Korea College of Medicine
- JHK@catholic.ac.kr
- Junseok Kwak
- SK Bioscience
- junnseokkwak@gmail.com
- Soo Ji Lee
- Celltrion Inc.
- sooji_0_0@hotmail.com
- Hyoungtae An
- Post-doc. Johns Hopkins University School of Medicine
- karin0297@korea.ac.kr
- Seungmin Yoo
- SPL Life Sciences
- smmagi@korea.ac.kr
- Juyeon Jeong
- Samsung Bioepis
- jjy2789@korea.ac.kr
- Jaeyeon Jung
- Reseacher
- jaeyeonie@korea.ac.kr
- Seukun Kim
- SK Bioscience
- yuha0607@korea.ac.kr
- Minsoo Kang
- Genexine
- lovesadly@korea.ac.kr
- Hyeryung Kang
JW C&C Research Laboratories
- hyeryung215@korea.ac.kr
- Yerin Jo
MD Biolab Co.
- joyelin33@naver.com
-
Jinseok Pyo
- Celltrion Inc.
- pyojs9320@korea.ac.kr
- Suhyun Kim
- dariakullen@korea.ac.kr
Beomsu Kim
- Samsung Bioepis
kjhh101@naver.com
Sangyun Kim
smallkid96@naver.com
Sila Han
sila.han@daum.net
